
Issue No. 148
Summarized from Journal of Clinical Periodontology, Volume 53, Issue 4, April 2026, 550-561
Editor: James Deschner, chair, EFP scientific affairs committee
Does adjunctive use of systemic amoxicillin and metronidazole improve outcomes of access-flap surgery for peri-implantitis?
Authors: Jarno Hakkers, Yvonne C. M. de Waal, Barzi Gareb, Henny J. A. Meijer, Gerry M. Raghoebar
Background
Peri-implantitis is a plaque-associated inflammatory disease affecting the tissues around dental implants, characterized by deep peri-implant pockets, bleeding and/or suppuration on probing, and progressive loss of supporting bone. Various non-surgical and surgical approaches have been proposed, but their long-term effectiveness is inconsistent and disease control remains challenging.
In the case of peri-implantitis, the peri-implant microbiome shifts towards a predominantly anaerobic, gram-negative community that includes both periodontal and site-specific pathogens. In periodontitis, adjunctive systemic amoxicillin and metronidazole in addition to mechanical debridement have shown synergistic antimicrobial effects and clinically relevant improvements, including reductions in red-complex organisms and Aggregatibacter actinomycetemcomitans.
On this basis, the same antibiotic regimen has been explored as an adjunct to peri-implantitis surgery. The available clinical studies show only modest and short-term improvements, and they differ in terms of decontamination protocols and outcome measures, which makes the overall benefit difficult to judge.
The EFP's clinical practice guideline on the prevention and treatment of peri-implant diseases emphasizes cautious use of systemic antibiotics and highlight the lack of robust long-term randomized controlled trials (RCTs) in the surgical treatment of peri-implantitis, leaving uncertainty about when, and for whom, such regimens are justified.
Aim
The aim of this investigator-blinded randomized controlled trial (RCT) was to evaluate the effect of adding a seven-day regimen of systemic amoxicillin and metronidazole to access-flap surgery for peri-implantitis with no-wall, one-wall, or two-wall bone defects, with a follow-up period of one year.
Materials and methods
- Study Design: Parallel-arm single-blinded RCT comparing access-flap surgery with systemic amoxicillin and metronidazole (test) with access-flap surgery alone (control).
- Participants:
- One hundred and twenty-three patients screened (January 2020 – August 2023, University Medical Centre, Groningen, Netherlands).
- Fifty-nine patients randomly assigned to test group (n=29) and control group (n=30)
- Diagnostic criteria (peri-implantitis):
- Bleeding (BOP) and/or suppuration on probing (SOP), increased probing pocket depth (PPD) compared to previous measurements, and bone loss beyond initial remodelling.
- If no baseline data: PPD ≥ 6mm + bone loss ≥3mm apical to the most coronal part of the intraosseous part of the implant.
- Inclusion:
- Persistent peri-implantitis after non-surgical submarginal instrumentation (NSI) with residual PPD >5mm and BOP/SOP.
- Included defects: no-, one-, and two-wall peri-implant bone defects.
- Pre-surgical interventions:
- Oral-hygiene instruction, professional cleaning.
- Mechanical debridement: hand instruments, air polishing (erythritol and 0.3% chlorhexidine).
- Surgical procedure:
- Full-thickness flap elevation, removal of granulation tissue with titanium curettes.
- Implant-surface decontamination: air polishing, 35% phosphoric acid (one minute) and saline rinse, final air polishing.
- Flap closure.
- Test group: amoxicillin + metronidazole (500 mg each), three times daily for seven days. Control group: no antibiotics.
- Outcomes:
- Primary outcomes: mean PPD, mean BOP.
- Secondary outcomes:
- Disease resolution (composite): PPD ≤5mm, no BOP, no SOP (based on the endpoints of surgical peri-implantitis therapy suggested by Herrera et al., 2023).
- SOP.
- Radiographic marginal bone level.
- Clinical and radiographic outcomes were reported at each timepoint at both implant and site level.
- Adverse events were recorded two weeks postoperatively.
- Follow-up timepoints: three, six, nine, and 12 months.
- Statistical analyses: mixed-effects models (linear and logistic) to evaluate the effects of the interventions on repeated measurements of the primary and secondary outcomes.
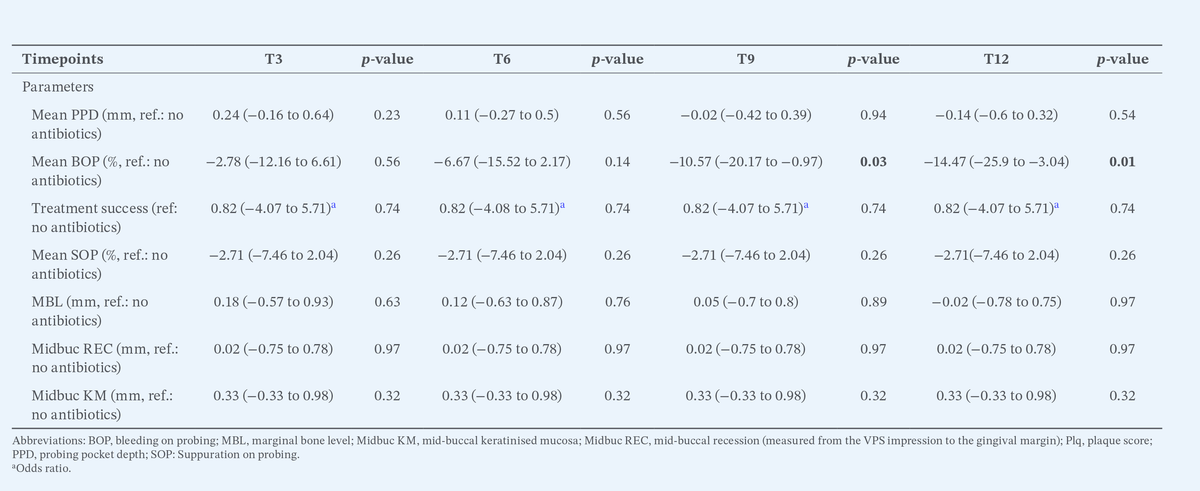
Results
- One patient in the test group and two patients in the control group discontinued the intervention. At the final timepoint, data was missing for one patient in the control group. Intention-to-treat (ITT) analyses were reported for 29 patients in the test and 30 patients in the control group.
- Baseline patient characteristics were similar in both groups. Notably, both groups contained a high proportion of smokers.
- At 12 months, mean BOP was significantly lower in the test vs. control group, with a difference of −14.47% (95% CI: −25.9 to −3.04; p = 0.01). Moreover, at the implant-level, the mean number of bleeding sites was significantly lower in the test vs. control group (2.3 + 1.9 vs. 3.5 + 1.8; p < 0.01). Similarly, a significantly lower proportion of implants presented with two or more bleeding sites in the test vs. control group (58.3% vs. 80.0%; p = 0.04).
- No significant differences were observed for any of the other parameters.
- Both groups reported few or no adverse events. Patients in the control group reported significantly (p < 0.05) greater pain, swelling of salivary glands, and teeth/protheses discoloration.
Limitations
- The one‑year follow‑up may be too short to capture long‑term recurrence and stability after peri‑implantitis surgery.
- The study was powered for continuous outcomes, not for binary “disease resolution”, leading to wide confidence intervals and limited precision for the composite endpoint.
- No microbiological analyses were performed, so potential microbiome‑level effects of antibiotics and their relationship to clinical response remain unexplored.
- The use of a fixed dual‑antibiotic regimen does not reflect individualised, culture‑guided therapy and may not be aligned with current principles of antibiotics stewardship.
- Inclusion of both dentate and edentulous patients could have affected results via differing oral microbiota but was not stratified in the main analyses.
Conclusions and Impact
- Adding systemic amoxicillin and metronidazole to access‑flap peri‑implantitis surgery produced a modest but statistically significant reduction in BOP at nine and 12 months.
- No additional benefit of antibiotics was observed for mean PPD, changes in marginal bone level, SOP, or the composite disease-resolution endpoint.
These results suggest that, for typical surgical peri‑implantitis cases, access‑flap debridement alone is usually sufficient, and routine addition of systemic amoxicillin and metronidazole is not justified.
Rapporteurs: Toma Betancur, Tonje Moen Eckhoff, Jenny Eikeng, Sunita Sharma, Marit Emanuelsen Selju, and Siddharth Shanbhag, supervised by Professor Odd Carsten Koldsland
Affiliation: Postgraduate programme in periodontology, University of Oslo, Norway
With kind permission from Wiley Online Library. Copyright © 1999-2026 John Wiley & Sons, Inc. All rights reserved




