Clinical & Translational Research, Systemic health, Article
Probiotics in treating periodontitis and diabetes: a more systemic than local effect?
13 May 2026
Probiotics are attracting increasing interest as an adjunct to periodontal therapy. Rocío Bujaldón Rodríguez, winner of the second prize for clinical research in the latest edition of the EFP’s annual Postgraduate Research Prize, describes a clinical trial that explored the effect of one probiotic on periodontal patients with diabetes.
Periodontitis and diabetes are two highly prevalent chronic diseases with a well-established bidirectional relationship. They share common pathophysiological mechanisms, particularly chronic low-grade inflammation, which plays a key role in the progression of periodontal diseases and in metabolic control. In this context, non-surgical periodontal treatment has been shown not only to improve clinical periodontal parameters but also to provide systemic benefits, including a reduction in glycated haemoglobin (HbA1c) of approximately 0.3–0.5%.
However, this treatment has limitations—such as the persistence of deep periodontal pockets and the rapid bacterial recolonization that occurs after therapy—which have driven the search for adjunctive strategies. Among these, probiotics have been attracting increasing attention. Probiotics are defined as live microorganisms that, when administered in adequate amounts, confer a health benefit on the host.
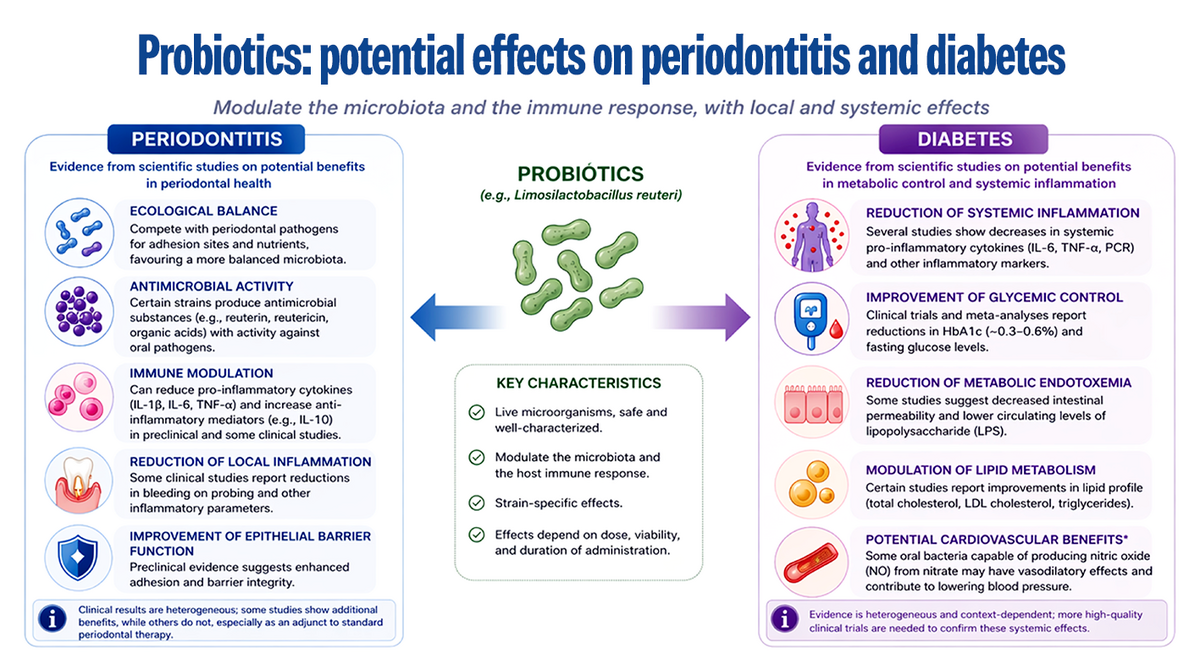
Their use reflects a broader conceptual shift in the understanding of periodontal diseases, which are now seen not so much as specific infections but as dysbiotic conditions involving complex microbial communities and an altered host response. From this perspective, probiotics are not intended to eliminate bacteria, but rather to modulate the microbial ecosystem and promote a balanced state.
Several mechanisms have been proposed to explain their potential effects. Certain probiotic strains may compete with periodontal pathogens for ecological niches, produce antimicrobial compounds, and interact with the host immune system. In particular, their immunomodulatory capacity—including the potential reduction of proinflammatory mediators—has been proposed as a relevant pathway.
This approach is especially relevant in patients with diabetes, as people with suboptimal glycaemic control tend to show a less favourable response to periodontal treatment, which is likely the result of an increased inflammatory burden and an altered immune response. In this context, strategies capable of modulating inflammation may have an impact not only at the periodontal level but also systemically. Nevertheless, translating these biological effects into consistent clinical benefits remains challenging.
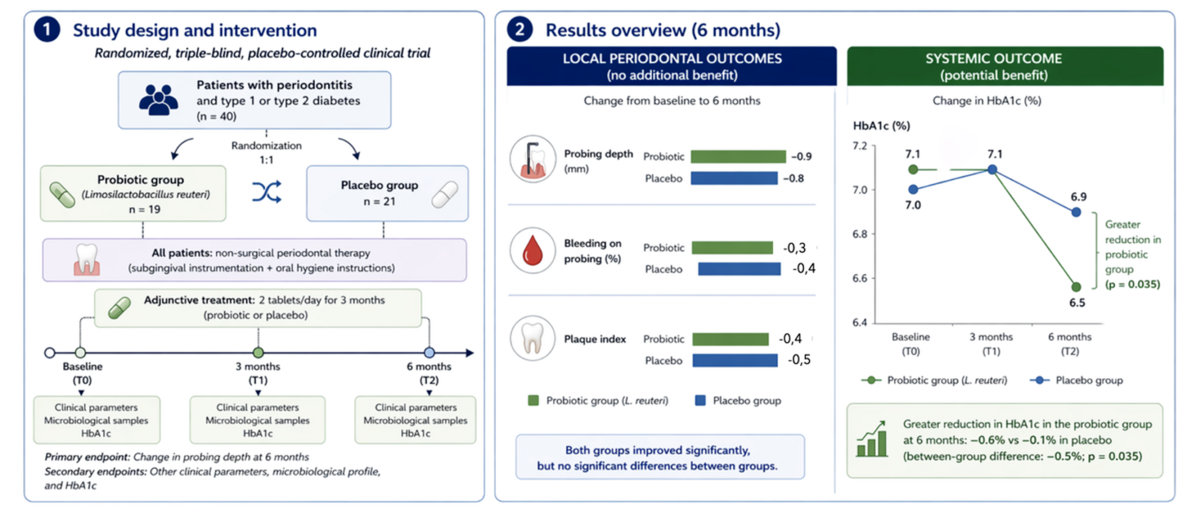
Our randomized, triple-blind, placebo-controlled clinical trial evaluated the use of Limosilactobacillus reuteri as an adjunct to subgingival instrumentation in patients with stage II/III periodontitis and type 1 or type 2 diabetes mellitus. After six months of follow-up, both groups showed significant clinical improvements following periodontal therapy. The mean probing depth reduction was 0.9mm in the probiotic group and 0.8mm in the placebo group, with no statistically significant differences between the groups. Likewise, no differential microbiological changes were observed.
However, when systemic outcomes were analysed, a different pattern emerged. Although both groups showed reductions in HbA1c levels, the decrease was greater in the probiotic group (0.6% versus 0.1% in the placebo group), and this difference of approximately 0.5% was statistically significant. This finding suggests that the effects of probiotics may not be primarily local but rather are mediated through systemic mechanisms.
For this reason, probiotics have been proposed to modulate systemic inflammation, influence the interaction between the oral and gut microbiomes, and contribute to metabolic regulation. In addition, some studies have shown that certain oral bacteria with nitrate-reducing capacity can contribute to the production of nitric oxide, a mediator with vasodilatory effects and a potential impact on blood pressure. However, these effects are species-specific and cannot be generalized to all probiotics.
This heterogeneity is a key aspect. The effects of probiotics are highly dependent on the strain, the host, and the clinical context. This may partly explain why additional periodontal benefits are difficult to demonstrate consistently, while systemic effects—particularly related to inflammation and metabolic control—are more frequently reported.
At present, probiotics should not be considered as a substitute for established periodontal therapies. However, they may represent a promising complementary strategy, particularly if we move from a purely local perspective to a more integrated view of oral and systemic health.
Biography

Rocío Bujaldón Rodríguez is a periodontist trained at the University of Granada and the Complutense University of Madrid (UCM), where she completed the EFP-accredited master’s programme in periodontology. She combines clinical practice in periodontology, oral surgery, and implant dentistry with research activity. Her main research interest is the relationship between periodontal and systemic diseases, which was the subject of her doctoral thesis.




