Prevention & Public health, Public health & Prevention, Article
Monitoring and controlling oral endotoxin levels for disease prevention
17 July 2023

In the second of three articles on the three winners of the EFP’s Innovation Award for Digital Solutions for Gum Health 2023, Perio Insight focuses on technology that would monitor and control oral endotoxin activity to enable personalised primary and secondary prevention of gum diseases and related systemic complications. Third-prize winner Svetislav Zaric, from King’s College London, UK, explains his innovation.
Most periodontal diseases develop insidiously, and it may take years for periodontitis to clinically manifest and to be diagnosed. So far, the diagnostic focus has been on determining the extent of the historical tissue breakdown – probing pocket depth (PPD) and clinical attachment loss (CAL) – rather than on detecting the biological start of the disease.
As periodontal diseases are episodic and site-specific in nature – with still-unknown factors that drive the progression from gingivitis to periodontitis – there are unique challenges in identifying and monitoring periodontal conditions. Different individuals demonstrate varying susceptibility to the onset and progression of periodontal diseases. Consequently, the application of uniform preventive and curative protocols will rarely meet individual needs, resulting in underprovision of care to some individuals and overprovision to others. This can lead to an increased disease burden, unwanted side effects, and the suboptimal allocation of resources, and so is an important issue for both primary and secondary prevention.
The provision of patient care guided by the assessment of patient-level and site-level risk for the development and progression of periodontitis may therefore be an advantageous approach. In order to determine the personalised, point-of-care treatment plan and to reduce healthcare costs and inequalities, there is a fundamental requirement for disease-specific biomarkers to distinguish progressive periodontitis from normal biological processes and to reveal hidden threats before the disease becomes clinically apparent.
The identification of patient-level and site-level biological signatures with the potential to highlight risk kinetics, to mark disease progression, to monitor health status, and to indicate treatment outcomes would be of significant clinical advantage. There is a need for the development of new, chair-side, point-of-time diagnostic tests that can detect the presence of active disease, predict future disease progression, and evaluate a patient’s response to periodontal therapy, thereby improving the clinical management of periodontal patients.
Moreover, epidemiological evidence accumulated over the last two decades suggests that periodontal diseases are associated with dyslipidaemia, glucose intolerance, hypertension, low-grade systemic inflammatory state, and with systemic diseases and conditions such as cardiovascular disease, diabetes, and obesity. Primary and secondary prevention of periodontal diseases is therefore important for alleviating the risk of these systemic complications and improving patients’ general wellbeing.
Molecular signatures
Our innovative approach focuses on the role of the oral microbiome’s molecular signatures in disease aetiology and pathogenesis, with the aim of developing a screening and monitoring tool for periodontal diseases and treating these conditions at or close to their biological onset. Specifically, our aim is to develop and validate a rapid and reliable digital-device testing protocol, based on the endotoxin profile of oral bacteria, which can be used routinely by dental-care professionals and the wider public to maintain and improve oral health and quality of life and to encourage access to dental care and reduce health disparities. The test would be made in two versions:
- Patients: Home salivary test, patient-performed self-testing for early detection and prevention of periodontal diseases or their relapse, before significant tissue destruction occurs.
- Oral-healthcare professionals: Chair-side test for subgingival plaque (Figure 1), for personalised, site-specific, point-of-time management of periodontal conditions.
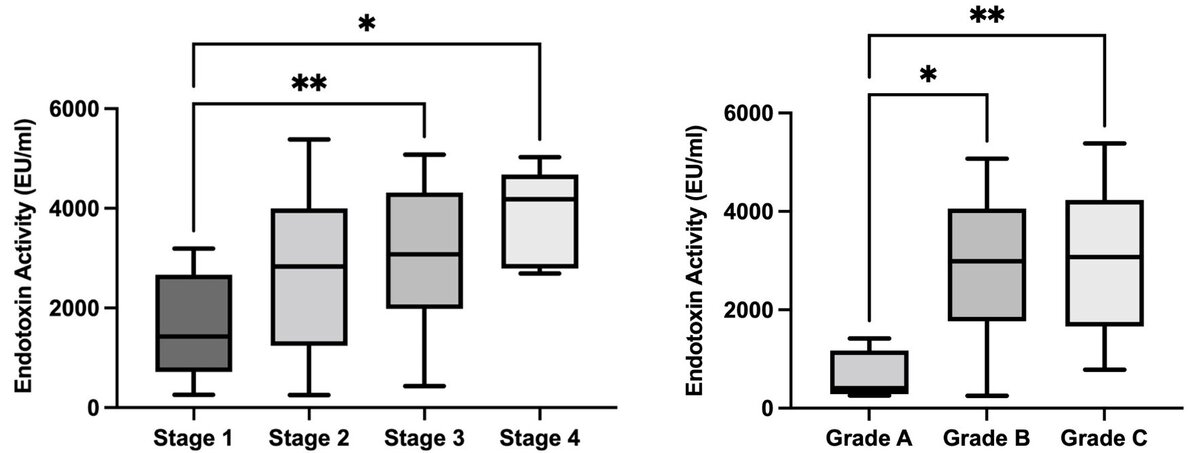
Endotoxin is a preserved moiety present only in Gram-negative bacteria, known for their potential to cause bone resorption directly, or indirectly through innate immunity-mediated inflammation. In addition, it is one of the mediators that links periodontal diseases with systemic conditions. Our test based on endotoxin activity can detect minor, initial dysbiotic changes in the oral environment that can lead disbalance between microbial challenge and the host immune response and eventually cause tissue damage. We have already shown significant correlations between subgingival endotoxin activities, probing pocket depth, and periodontal diagnoses (according to the stages and grades of the 2018 classification of periodontal diseases), which were independent of patients' age, gender, and smoking status (Figure 2)
This proposal aims to radically change clinical periodontal practice from reactive to personalised and preventive, and treatment from non-selective and mechanical to pharmacologically precise. A patient’s susceptibility would be determined and monitored before any clinical manifestations of the disease and specific bacterial virulence factors – such as lipopolysaccharides (LPS) – could thus be precisely neutralised.

Biography
Svetislav Zaric is an NIHR Academic Clinical Lecturer and a Specialty Registrar in Periodontology, at the Faculty of Dentistry, Oral & Craniofacial Sciences, Centre for Host-Microbiome Interactions, King’s College London and Guy’s Hospital, United Kingdom. He obtained his primary dental degree and a master’s degree from University of Belgrade, Dental School in Serbia and his doctorate in host-pathogen interactions and mechanisms for the control of chronic inflammation from Queen’s University Belfast in Northern Ireland. His current research focus is on translational sciences linking microbial-associated molecular patterns and pathogenesis of periodontal diseases, together with the discovery of new mechanisms for decreasing the microbial burden and limiting the host inflammatory response.




