Prevention & Public health, Systemic health, Article
How an AI-based mobile app can tackle low-grade systemic inflammation
29 March 2023
Low-grade systemic inflammation is known as the “silent killer” and there is a clear association with periodontal disease. Modifiable lifestyle factors —including diet, smoking, exercise, and oral hygiene — play a fundamental role. In this context, encouraging people to adopt healthy lifestyles can help prevent disease and improve health. Italian researchers Mario Aimetti and Giacomo Baima explain the mobile application they are helping to develop, which uses artificial intelligence to encourage users to make lifestyle changes that will benefit both their gum and their systemic health.
Inflammation is a response mechanism that our biological systems use to cope with injury and disease. We are constantly exposed to numerous triggers — such as trauma, infections, and metabolic challenges — that can elicit an inflammatory reaction.
While an acute inflammatory reaction is essential to the body’s healing processes, a prolonged response can wear down the immune system. This chronic condition, known as low-grade systemic inflammation (LGSI), occurs when the trigger cannot be switched off or when the body loses its ability to resolve inflammation after repeated stressors.
Scientific discoveries over the past two decades have reinforced the belief that LGSI causes the body to produce inflammation continuously, even in the absence of the initial eliciting trigger. This long-lasting inflammatory activity disrupts immunological tolerance, can alter tissues and organs, and increases the overall risk of non-communicable diseases (NCDs).
Inflammation-related NCDs such as cardiovascular diseases, chronic kidney disease, cancer, and type-2 diabetes mellitus account for more than half of all deaths worldwide, so LGSI is a significant contributor to disability and mortality. Because of these well-documented effects, LGSI is often referred to as the "silent killer”.
LGSI and periodontitis
Periodontitis affects up to 50% of the worldwide population — it is the sixth most common NCD — and it is associated with a broad spectrum of systemic diseases via the induction of LGSI. Current evidence points to a bidirectional relationship between periodontitis and LGSI. Periodontal infection causes a spill-out of bacterial toxins and inflammatory mediators that induce or sustain the chronic systemic inflammatory milieu. Looking at things from the other direction, it is also plausible that LGSI mediates immune-system dysfunctions that may predispose an individual to increased susceptibility to the onset and progression of periodontitis.
Various inflammatory biomarkers, such as pro-inflammatory cytokines or acute-phase reactants present in the blood at different levels, can help identify LGSI in an individual. Among these, the acute-phase reactant C-reactive protein (CRP) is one of the most reliable and frequently tested inflammatory biomarkers that can confirm the presence of LGSI. However, only high-sensitivity CRP (hsCRP) tests can accurately assess CRP levels. The American Heart Association and the Centers for Disease Control and Prevention have established hsCRP cut-offs of 1mg/L for moderate risk and 3mg/L for high risk of hsCRP elevation, which are synonymous with LGSI (Figure 1).
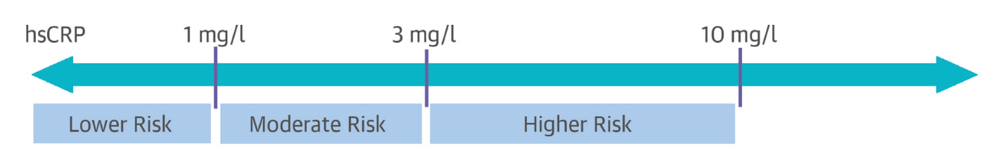
Other LGSI biomarkers include blood cellular markers, such as cytokines and chemokines, total leucocytes, platelets, adipokines, and acute-phase proteins. However, they have not yet been investigated as extensively as CRP and they have not been as consistently and reliably linked to states of chronic disease.
Scientific evidence therefore indicates that hsCRP is probably the most reliable single biomarker of LGSI and the best predictor of cardiovascular and all-cause death. Indeed, much research has shown how periodontitis is associated with high hsCRP levels and a hyperinflammatory systemic status, whereas periodontal treatment has been consistently related to a decrease in the levels of LGSI markers in blood, saliva, and gingival crevicular fluid.
LGSI has been linked to unhealthy lifestyle choices
Most LGSI risk factors are tied to environmental, lifestyle, and biological factors — which means that they are preventable. Unhealthy diet and alcohol abuse, weight gain, physical inactivity, psychological stress, lack of sleep, environment (e.g., smoking, air pollution, or exposure to chemical contaminants), and gum health are among the modifiable risk factors for LGSI.
Lowering LGSI levels by altering these risk variables can reduce the risk of non-communicable diseases (NCDs) and overall mortality. It is encouraging to know that simple changes in daily routines and individual behaviour can achieve this positive effect. Additionally, the best way to avoid the onset of LGSI and its associated disorders is through early preventive interventions. Implementing this change requires a personalised and focused strategy that considers each individual’s life context.
It is widely known that eating well, being physically active, and brushing teeth properly are key elements in preventing or managing local and systemic inflammation. Unfortunately, these healthy behaviours are frequently neglected. To provide personalised healthcare interventions on a large scale, eHealth (electronic health) strategies offer an effective solution through increased access to educational information, patient health data, and powerful data analytics.
Artificial intelligence (AI) has the potential to revolutionise tailored lifestyle interventions and, with the rapid advancement of mobile technology, AI-based platforms can be developed to help individuals manage their health and wellbeing. The ability of an AI-based mobile application to gradually learn from its users is one of its main features and allows continuously updated recommendations based on the user’s progress. This enables a more dynamic and adaptable strategy for individualised lifestyle interventions.
In this context, the research department of Swiss company Shealthy World* is seeking to raise awareness of low-grade chronic systemic inflammation and its global health consequences, and to help people assess, monitor, and lower their LGSI status in a non-invasive way through a scientifically sound model.
A multidisciplinary team of scientists and technologists from around the world is developing a digital platform that will allow users to be profiled via a digital patient history to assess inflammation-relevant aspects of their lifestyle (Figure 2). The questionnaire requests information relating to the user’s habits in four areas — nutrition, movement, cognition, and environment — within a holistic approach. The answers given by the user will be processed to calculate a personalised inflammatory score, using a proprietary algorithm based on AI and machine learning.
The platform can use data from various sources — such as wearable devices, mobile health apps, and electronic health records — to facilitate the user’s experience and limit the number of profiling questions. It can then deliver personalised tips to change or improve habits in nutrition, movement, cognition, and environmental factors, while also providing relevant in-depth text and scientific references. All user-generated content and data is pseudo-anonymised, in accordance with the European Union’s General Data Protection Regulation and its advisory Article 29 Working Party (Art. 29 WP).
The mobile application will incorporate gamification and reward elements to enhance user engagement and motivation. Gamification entails integrating game components into non-game settings to entice users with appealing features and infuse some fun into otherwise monotonous tasks while also offering motivational and cognitive advantages.
Research has shown that gamification can help address the gap in healthcare resources and information access by making use of rewards and feedback. In the context of our AI-based mobile application, rewards may be linked to specific industry partners, while gamification elements could consist of a personalised interactive avatar that proposes easy-to-achieve objectives to maintain motivation and drive outcomes. By incorporating games and rewards, the application can help increase users’ adherence to the recommended lifestyle changes.
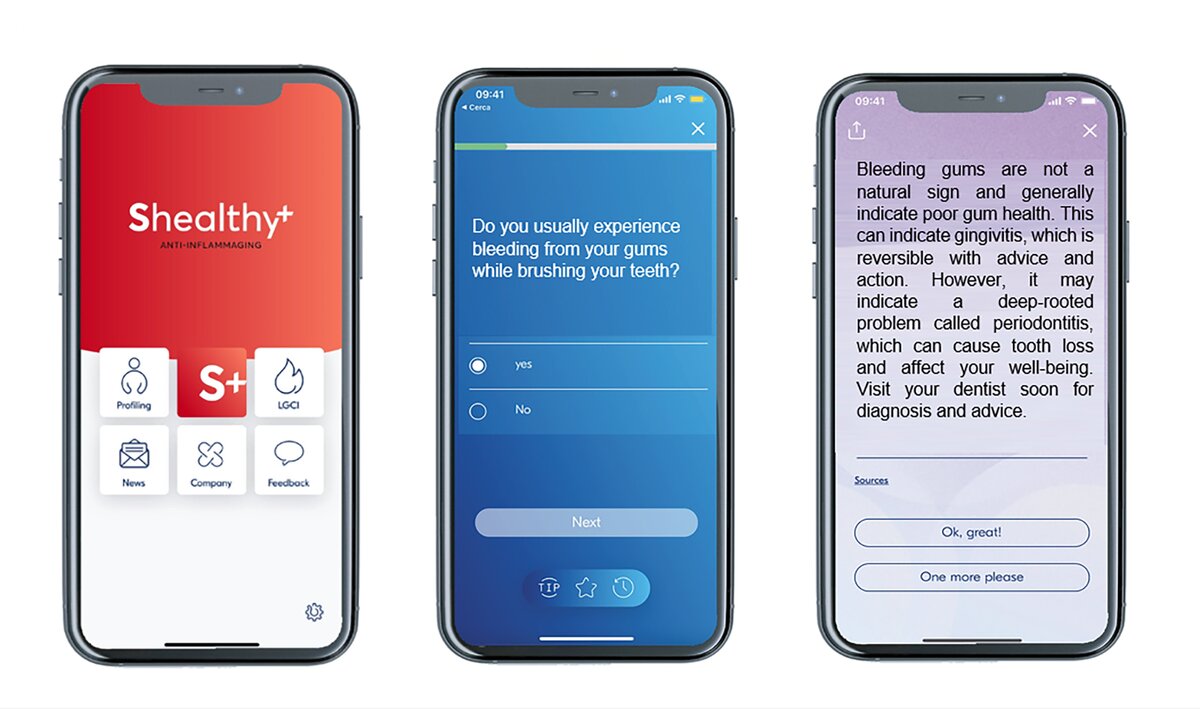
The personalised inflammatory score provided by the app — using a patented aging (AGI) index — is developed from the US National Health and Nutrition Examination Study (NHANES) public dataset and peer-reviewed publications that have examined the associations between various parameters and hsCRP. Scientific papers were selected with the following order of priority:
- meta-analyses, including both observational studies and clinical trials.
- meta-analyses, including observational studies or clinical trials.
- cohort-based observational studies.
- clinical trials or other published studies with a lower number of participants.
In evaluating the score, two components are considered: statistical inflammatory potential and the personal level of specific parameters (e.g., quantification of total physical activity, dietary intake of certain foods), both of which can vary significantly. Based on these two components, the values found for the influence of a specific parameter on the hsCRP will be weighted to standardise the data collected from different studies and from the NHANES dataset.
The index also offers dynamic variability through the tracking of physical activity, sleep quality, and heart-rate variations, as well as the questionnaires that the AI algorithm will assess with non-monitorable information (such as eating habits). The aim is to achieve user-friendly data collection, thereby reducing manual data input to the bare minimum.
Implementation of the AI mobile application for gum health
To further expand the potential benefits of the AI-based platform, it could be tailored for use by dental professionals to provide to their patients. This implementation would have two main aspects. First, by looking at periodontal infection as one of the main drivers of LGSI induction, primary and surrogate signs of periodontitis can enter the AI algorithm for the overall risk assessment of LGSI. Second, the AI-based mobile application could have a significant impact on the prevention and management of gum diseases, as well as other NCDs.

Thanks to the impact of receiving one of the prizes in last year’s EFP Innovation Award for Digital Solutions for Gum Health, our research team at the University of Turin is working on the implementation of the gum-health section of the AI-based mobile app software that is being developed by Shealthy World. Traditional treatments are often reactive rather than proactive and may not address the underlying causes of the disease. This is where AI-based mobile applications come in and can help dental professionals in their therapy. In fact, the app could be used to gather information on an individual’s oral-hygiene practices, smoking, diet, physical activity, and other determinants for periodontitis risk.
The software can then offer the user specific advice on how to maintain better oral hygiene in the context of the primary prevention of periodontitis. It can also prompt the user to schedule routine dental check-ups and cleanings, which can aid in secondary and tertiary prevention. For instance, the app can track the user’s oral- hygiene habits — such as brushing and flossing frequency — and provide instant feedback on their progress. It can also provide personalised tips based on the user's oral-health status. For example, the app can recommend foods that are high in antioxidants and vitamins C, D, and E to help strengthen the teeth and gums.
The potential for the implementation of eHealth in the daily life of our patients is tremendous, but efforts still need to be made to avoid marketing-driven tips and stick to evidence-based recommendations. The idea is to use only science-based tips validated by a multidisciplinary independent team of experts and scientists, who have agreed to collaborate with the company. We hope that our future contribution can unlock new scientific opportunities to place oral and gum health at the gateway of systemic inflammatory control, with the final goal of improving our patients’ health and extending their life span.
* The authors declare that they have no commercial relationship with Shealthy World.
Select bibliography
Baima G, Romandini M, Citterio F, Romano F, Aimetti M. 2022. Periodontitis and Accelerated Biological Aging: A Geroscience Approach. J Dent Res. 101(2):125–132.
D’Aiuto F, Gkranias N, Bhowruth D, et al. Systemic effects of periodontitis treatment in patients with type 2 diabetes. Lancet Diabetes & Endocrinol. 2018.
Dugas M, Gao GG, Agarwal R. Unpacking mHealth interventions: A systematic review of behavior change techniques used in randomized controlled trials assessing mHealth effectiveness. Digit Health. 2020 Feb 20;6:2055207620905411.
Fedewa MV, Hathaway ED, Ward-Ritacco CL. Effect of exercise training on C reactive protein: a systematic review and meta-analysis of randomised and non-randomised controlled trials. Br J Sports Med. 2017; 51:670–6.
Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol. 2018; 15:505–22.
Furman D, Campisi J, Verdin E, Carrera-Bastos P, Targ S, Franceschi C et al. Chronic inflammation in the etiology of disease across the life span. Nat Med. 2019; 25:1822–32.
Hajishengallis G, Chavakis T. Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nat Rev Immunol. 2021 Jul;21(7):426-440.
Marruganti C, Baima G, Grandini S, Graziani F, Aimetti M, Sanz M, Romandini M. Leisure-time and occupational physical activity demonstrate divergent associations with periodontitis: A population-based study. J Clin Periodontol. 2023. Epub ahead of print.
Marsland AL, Walsh C, Lockwood K, John-Henderson NA. The effects of acute psychological stress on circulating and stimulated inflammatory markers: A systematic review and meta-analysis. Brain Behav Immun. 2017; 64:208–19.
Minihane AM, Vinoy S, Russell WR, Baka A, Roche HM, Tuohy KM et al. Low-grade inflammation, diet composition and health: current research evidence and its translation. Br J Nutr. 2015; 114:999–1012.
National Center for Health Statistics. NHANES questionnaires, datasets, and related documentation. 2020. wwwn.cdc.gov/Nchs/Nhanes.
Irwin MR, Olmstead R, Carroll JE. sleep disturbance, sleep duration, and inflammation: a systematic review and meta-analysis of cohort studies and experimental sleep deprivation. Biol Psychiatry. 2016; 80:40–52.
Pedersen BK. Anti-inflammatory effects of exercise: role in diabetes and cardiovascular disease. Eur J Clin Invest. 2017; 47:600–11.
Sardi L, Idri A, Fernández-Alemán JL. A systematic review of gamification in e-Health. J Biomed Inform. 2017 Jul;71:31-48.
Biographies

Giacomo Baima received his DDS from the University of Turin (Italy) in 2016 and then an MSc from the Complutense University of Madrid (Spain), before returning to Turn for his postgraduate training in periodontology under the mentorship of Prof. Mario Aimetti, achieving EFP board accreditation in 2022. He is currently a PhD candidate in bioengineering and surgical sciences and a research fellow at the University of Turin’s periodontology section, with a full-time involvement in clinical and basic research in periodontology and peri-implant diseases. He has authored various articles in international peer-reviewed journals and is a member of the EFP Alumni board.

Mario Aimetti obtained the Degree in Medicine and Surgery with honours from the University of Turin (1987) and a speciality in dentistry (1990) and maxillofacial surgery (1995). He is head of the periodontology department and director of the EFP-accredited postgraduate programme in periodontology at the University of Turin. A past president of the Italian Society of Periodontology and Implantology (SIdP) and president of SIdP Foundation, he is a member of the EFP’s education committee (2023-2026). Prof. Aimetti is the author of 130 articles published in international peer-reviewed journals and an international speaker.




