![]()
8 June 2023
EFP publishes guideline on the prevention and treatment of peri-implant diseases
Categories:Clinical Practice, Publications
The EFP guideline on "Prevention and treatment of peri‐implant diseases—The EFP S3 level clinical practice guideline" is now online in the Journal of Clinical Periodontology.
A special issue, that will include the guideline document as well as the associated systematic reviews, will be published soon.
This guideline follows the two recently published Clinical Practice Guidelines (CPGs) for the treatment of stages I-III and stage IV periodontitis that provided evidence- and consensus-based clinical recommendations following the high quality S3 approach, implementing the 2018 classification scheme.
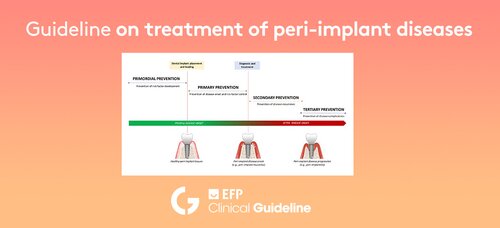
The new guideline focuses on the prevention and treatment of peri-implant diseases, delivering consistent, interdisciplinary, and evidence-based approaches, required to prevent the development of peri-implant diseases or their recurrence, and to treat patients with dental implants following the development of peri-implant diseases.
The CPG aims to guide oral health professionals and stakeholders such as other medical professionals, health systems, policymakers, patients and, indirectly, the public on the most effective available modalities to maintain healthy peri-implant tissues, and to manage peri-implant diseases.
The guideline states that prevention of peri-implant diseases should commence as soon as dental implants are planned, and later when they are surgically placed and prosthetically loaded. Once the implants are loaded and in function, a supportive peri-implant care programme should be structured, including periodical assessment of peri-implant tissue health. If peri-implant mucositis or peri-implantitis are detected, appropriate treatments for their management must be rendered.
“Peri-implant diseases, specifically peri-implant mucositis and peri-implantitis, are highly prevalent and their management is challenging,” says David Herrera, first author of the publication. “This clinical practice guideline provides professionals with advice on the management of peri-implant diseases. The recommendations have been formulated following a rigorous evidence-based and patient-centred decision-making process.”
EFP clinical practice guidelines are developed following methodological guidance from the Guidelines International Network, the Association of Scientific Medical Societies in Germany and the Grading of Recommendations Assessment, Development and Evaluation (GRADE) process. A rigorous and transparent process includes synthesis of relevant research in 13 specifically commissioned systematic reviews, evaluation of the quality and strength of evidence, formulation of specific recommendations, and a structured consensus process involving leading experts and a broad base of stakeholders.
Moritz Kebschull, co-author of the publication adds: “Peri-implant diseases are a growing public health problem due to their high occurrence and associated consequences, including substantial dental care costs. Following the recommendations will facilitate a consistent, interdisciplinary, and evidence-based approach to the prevention and treatment of peri-implant diseases.”




